Types of enzymes and their functions
The interference resulting from the simple fact oxygen binding proteins, hemoglobin and myoglobin, possess precisely the exact endothelial structure and also execute precisely the very same purposes, yet captured coffins once when it had been discovered that the comparable situation does occur in instance of mammalian serine proteases.
These enzymes have been termed since they’ve some exceptional serine deposits that react irreversibly with organ phosphorus chemicals, by way of instance, using isopropyl fluorophosphates. Their task includes an optimal in u and can be dependent on the ionization condition of their classes.
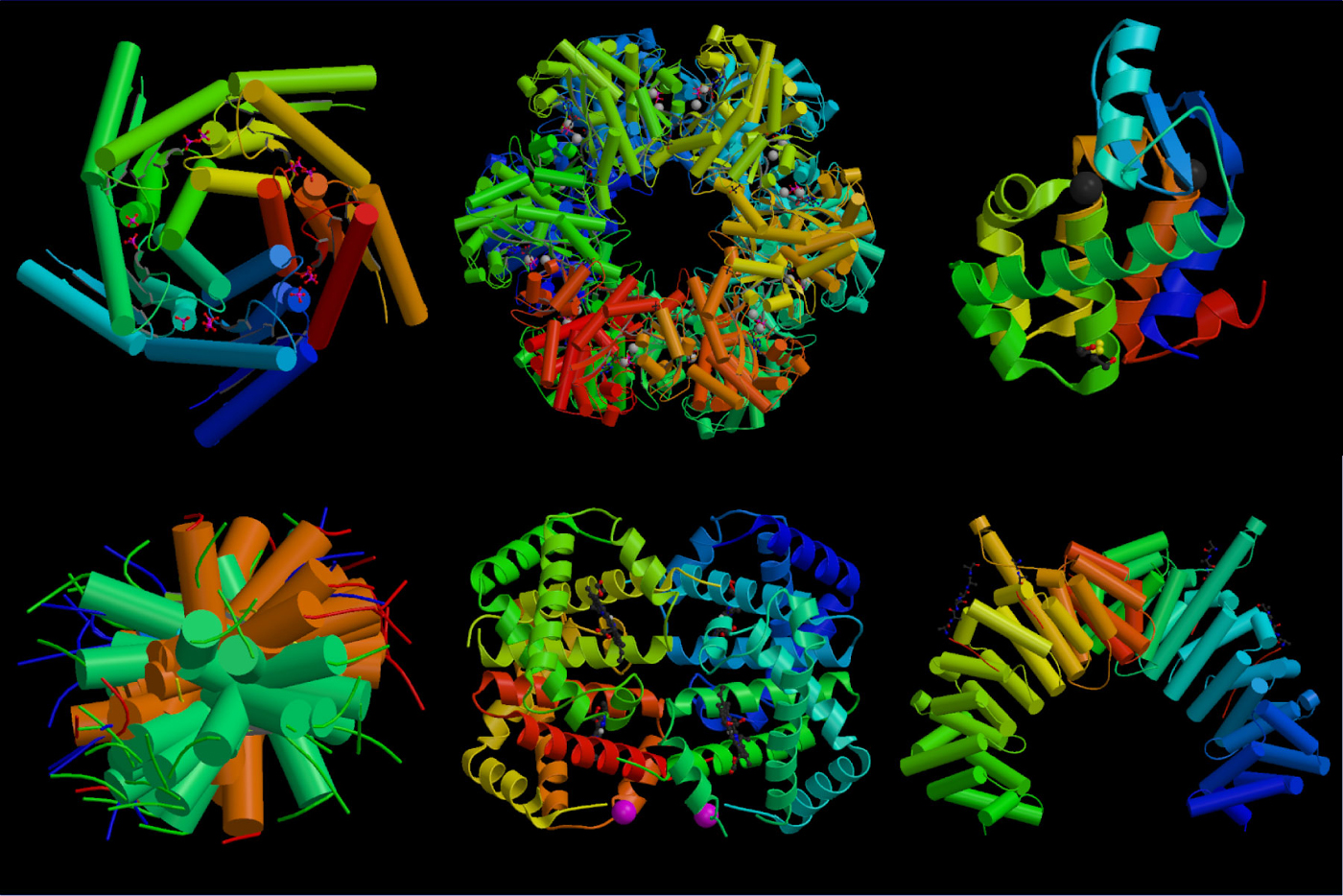
In every 3 circumstances an “acyl enzyme” is shaped throughout the response, at the carboxyl fragment of this substrate types a ester bond with the hydroxyl number of their busy serine. At the molecule of chymotrypsin, there is a well-defined pocket connecting significant hydrophobic side chains.
The negatively charged carboxyl group sorts an ionic bond with absolutely charged ammonium or classes at the end of the lysine or arginine chain. Two glycine found in the entry to this wallet of chymotrypsin, in example of lactase, are replaced by valise and threonine. It stops the insight of big side chains in to the pocket and also ensures the binding of the small side chain of alanine.
The conclusion of this arrangement of crystalline enzymes demonstrated the polypeptide backbones of most 3 enzymes have been almost overlapping with each other, other than for all areas where by a few proteins are lost or added. The gap within the specificity of the enzymes is caused to little fluctuations from the arrangement of this “wallet” that hardens the opposite chain of this amino acid.
At the molecule of chymotrypsin, there’s really a well-defined pocket linking huge hydrophobic side chains. Two glycine positioned in the entry to this wallet of chymotrypsin, within instance of elastic, are changed with valise and threonine. It stops the insight of big side strings in to the wallet and also guarantees that the binding of the tiny side chain of alanine.
The striking part of this endothelial arrangement of the enzymes shared previously might be anticipated by the contrast to this 6 amino acid sequences. Between your most important structures of the enzymes can be just a somewhat higher homology, nevertheless the amino acid makeup of elastic and chymotrypsin participates with all the Amino acid structure of trypsin by just 50 percent.