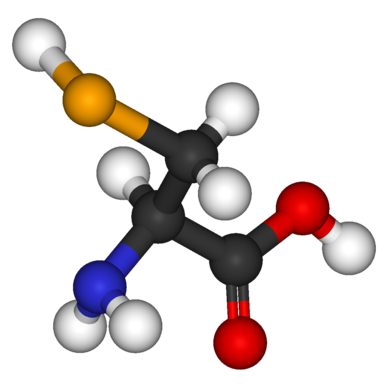
Specificity and relative reactivity of enzymes
Occasionally it’s likely to instantly explore Active enzyme substrate intricate below situations where the enzymatic response doesn’t move. These processes derive using gradually responding substrates in pH as soon as the enzyme is all but entirely uncalled as an effect of the changeover of this rest of the catalytic facility to some other ionic condition or at incredibly lower temperatures or even with a chemically altered enzyme.
A typical case of a compound alteration may be that the loss of the carboxyl set of the catalytically significant deposits of lysozyme to an alcohol set. Sometimes it’s likely to instantly explore Active enzyme substrate intricate having a quick response. This potential is comprehended in many cases as soon as equilibrium is based on the substrate and also this item, hidden in the creation of the substrate. The accession of the enzyme into this balance mix cannot alter the balance status while in the process, being a consequence that it really can get a well-balanced enzyme substrate intricate.
But there’s absolutely not any certainty which the balance posture that does occur from the process is still exactly the exact same to related reagents. A good illustration of this kind of balance could be that the hydrolysis of the peptide, favored synthesis that is aroused by inserting a surplus of amine. This strategy has been also utilized in experiments using all application.
The balance will be changed in the creation of just one of these kinds. Remember that because of its inverse response, the enzyme product elaborate is an increasingly enzyme substrate intricate.
Cases of this application form of most of these procedures have been shared. Under, the serine proteases and also lysozyme are shared at greater depth. All these enzymes also have performed a necessary part within the maturation of these suggestions and processes talked about here and also therefore are usually discussed inside this novel. To begin with, being an effect of the assault of this substrate using hydroxyl, a covalently sure reciprocal is shaped, that turns out to a tetrahedral intermediate.
Crystallographic research also has made it feasible to set up the arrangement of nearly all of those complexes. Nature has presented us with an uncommon possibility to set up the arrangement of enzyme substrate part of trypsin and chymotrypsin using polypeptides, developing a assortment of polypeptide inhibitors which bind very closely to trypsin and chymotrypsin, as they’ve been mended from the conformation the substrate carries up on binding.