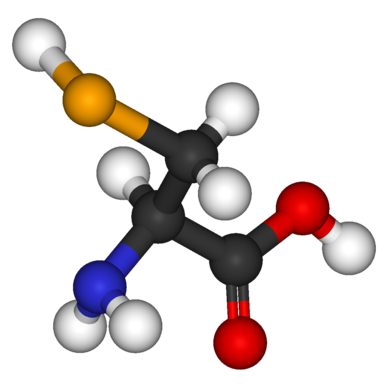
Building the name of the organic compound
Define the name of the main chain and form the ending depending on the older functional group. The degree of saturation of the main chain is reflected by suffixes: an – saturated carbon skeleton, en – the presence of double and in – triple bonds. Define the name of the substitutes – the younger functional groups, hydrocarbon radicals, which are designated by prefixes in a single alphabetical order.
A digit corresponding to the number of the carbon atom to which the substituent is bonded indicates the position of each substituent and each multiple bond. For a multiple bond, indicate the smallest number of the carbon atom at which this bond is located. The numbers are placed before the prefixes and after the suffixes or endings.
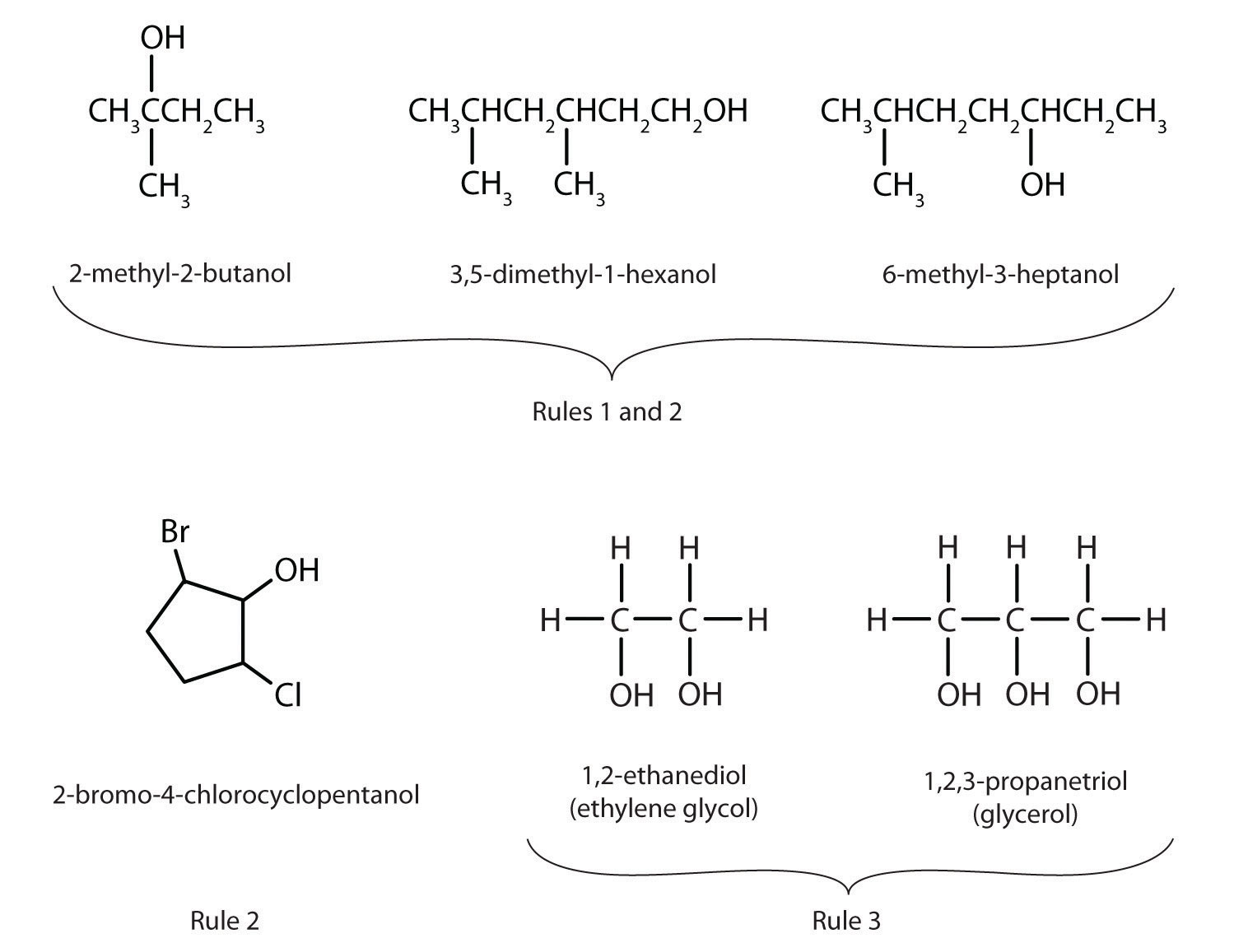
If the compound has several identical substituents or multiple bonds, then before the corresponding designation a multiplying prefix is placed: di, tri, tetra, etc.
Below are some examples of names for the IUPAC substitution nomenclature.
Radical-functional nomenclature
The use of the radical-functional nomenclature is more limited than the substitutionary nomenclature. It is mainly used for the name of the simplest mono- and bifunctional compounds and certain classes of natural substances.
Radical-functional nomenclature largely resembles and replaces the previously used rational nomenclature. Below are some examples of the names of organic compounds by the radical functional nomenclature.
In case of more complex compounds selected arbitrarily main chain indicate substituents in consoles, and their location is indicated by numerals prefixes ortho-, meta-, para- or Greek letters α, β, γ, δ, etc.
The stated nomenclature rules should serve as a permanent guide for the subsequent study of the nomenclature of specific classes of organic compounds and their individual representatives. Knowledge of the general rules of the nomenclature is of great importance, since according to them the names of numerous medicines are being built, with which doctors will constantly deal in their professional activities.